- Blog
- Avast cleanup coupon
- Twin mirror ending
- Schick coupons
- Transformice totems
- Icalamus 2 manual
- Ammonite tattoo
- Reading level the green ember
- Barney clark
- Phoenix creature
- Chromecast com setup
- Osirix md sorting
- Paranoid personality disorder treatment
- Faceless menace ffoil legendaries
- Theme park tycoon 2 ideas aesthetic
- A perfect day danny cope
- Puzzle dimension trophy guide
- Icopy bot reviews
- Popcorn time type
- Zombie driver summer of slaughter
- Download zapya for windows 7
- Colormunki display vs spyder 3 pro
- Hik vision darkfighter varifocal lens
- Wordpress for mac install
- The dreamfall chapters
- Engineered protein scaffold inhibitor
- Blog
- Avast cleanup coupon
- Twin mirror ending
- Schick coupons
- Transformice totems
- Icalamus 2 manual
- Ammonite tattoo
- Reading level the green ember
- Barney clark
- Phoenix creature
- Chromecast com setup
- Osirix md sorting
- Paranoid personality disorder treatment
- Faceless menace ffoil legendaries
- Theme park tycoon 2 ideas aesthetic
- A perfect day danny cope
- Puzzle dimension trophy guide
- Icopy bot reviews
- Popcorn time type
- Zombie driver summer of slaughter
- Download zapya for windows 7
- Colormunki display vs spyder 3 pro
- Hik vision darkfighter varifocal lens
- Wordpress for mac install
- The dreamfall chapters
- Engineered protein scaffold inhibitor
however, the inclusion of a 1:1 molar ratio of wild-type PFD inhibited CS. Here we review different classes of engineered proteins for their binding epitope geometry and as leads for synthetic secondary and tertiary structure mimics. Engineering Ultrastable Protein Scaffold for the Controlled Assembly of. The advantage of focused screens with conformationally-defined protein scaffolds is that conservation of the geometry of the natural binding epitopes may preserve binding site specificity while allowing direct mimicry by various synthetic secondary structure scaffolds. However, direct mimicry often does not lead to high affinity ligands because the natural complexes themselves are functionally transient and.

Rationally designed protein-protein interaction inhibitors mimic interfacial binding epitopes, specifically residues that contribute significantly to binding. Bode W, Huber R (1992) Natural protein proteinase inhibitors and their. Engineered protein scaffolds as leads for synthetic inhibitors of protein-protein interactions. Engineered proteins displaying conformationally-defined epitopes may serve as attractive alternatives to natural protein partners as they can be strictly screened for tight binding. Here we review different classes of engineered proteins for their binding epitope geometry and as leads for synthetic secondary and tertiary structure mimics. New binding protein scaffolds are constantly being designed or discovered with. Early examples were the Affibody, Monobody (Adnectin), and Anticalin proteins, which were derived from fragments of streptococcal protein A, from the tenth type III domain of human fibronectin, and from natural lipocalin proteins. The mimics typically need to be optimized for potency. The concept of engineering robust protein scaffolds for novel binding functions emerged 20 years ago, one decade after the advent of recombinant antibody technology.
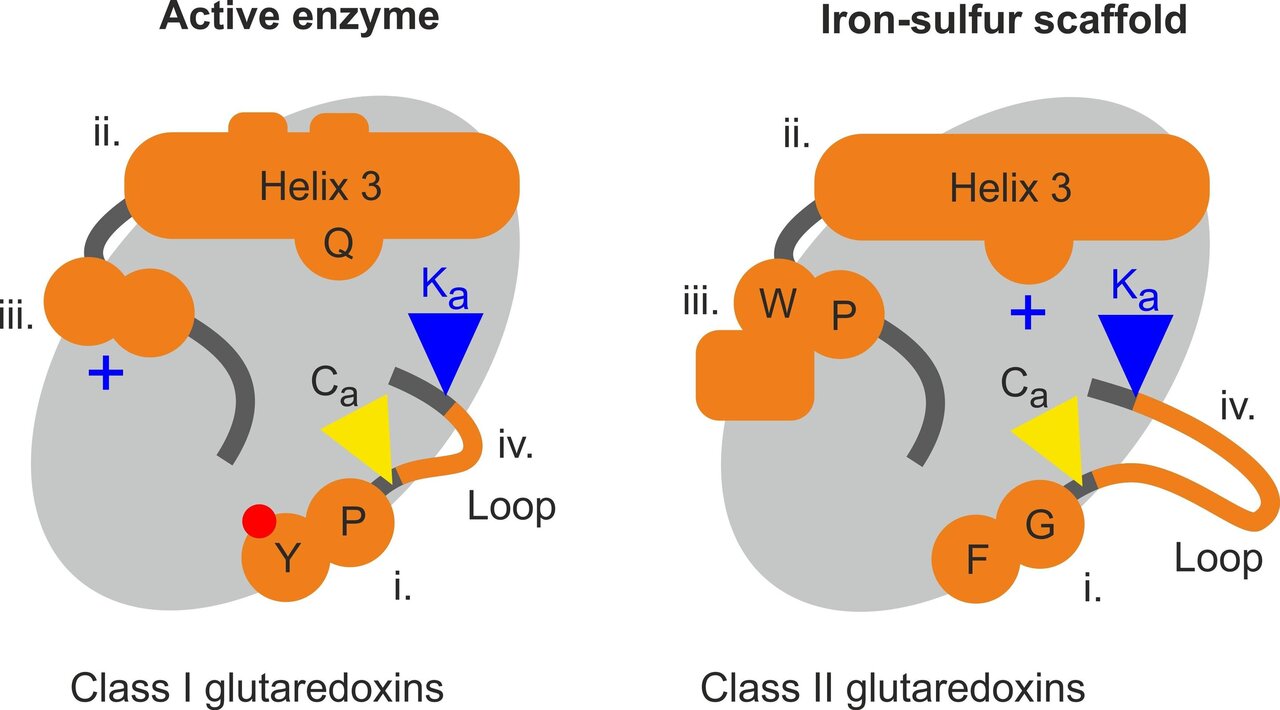
However, direct mimicry often does not lead to high affinity ligands because the natural complexes themselves are functionally transient and of low affinity. Rationally designed protein–protein interaction inhibitors mimic interfacial binding epitopes, specifically residues that contribute significantly to binding.
